 These species were deposited on a cryogenically cooled infrared transmissive window at 15 K. Fourier transform transmission spectroscopy (500–4000 cm −1/20–2.5 μm, 1.0 cm −1 resolution) was used to investigate solid acetaldehyde, ethanol and dimethyl ether, pure or mixed with water, CO, methanol, or CO:methanol. 
This work provides infrared reference spectra of acetaldehyde (CH 3CHO), ethanol (CH 3CH 2OH), and dimethyl ether (CH 3OCH 3) recorded in a variety of ice environments and for astronomically relevant temperatures, as needed to guide or interpret astronomical observations, specifically for upcoming James Webb Space Telescope observations. Accurate spectroscopic laboratory data of frozen COMs, embedded in ice matrices containing ingredients related to their formation scheme, are still largely lacking.Īim. At present only smaller molecular species have been directly identified in space in the solid state. Recent laboratory studies show that many such species form on icy dust grains. The number of identified complex organic molecules (COMs) in inter- and circumstellar gas-phase environments is steadily increasing. Max-Planck Institut für Extraterrestrische Physik (MPE),Ĭontext. Institute for Astronomy, University of Hawaii, Universities Space Research Association, Stratospheric Observatory for Infrared Astronomy, NASA Ames Research Center, Raymond and Beverly Sackler Laboratory for Astrophysics, Leiden Observatory, Leiden University,Į-mail: Observatory, Leiden University, Astronomical objects: linking to databases.Including author names using non-Roman alphabets.Suggested resources for more tips on language editing in the sciences Punctuation and style concerns regarding equations, figures, tables, and footnotes This is usually true unless the alcohol is exceptionally pure. Notice that the alcohol proton is not involved in spin-spin splitting. The position of the -OH peak can vary depending on the conditions, such as the NMR solvent used, the concentration of the alcohol, the purity of the alcohol, temperature, and if any water is present in the sample. These peaks tend to appear as short, broad singlets. Protons directly attached to the alcohol oxygen often appear in the region of 2.0 to 2.5 ppm.The electronegativity of the alcohol oxygen de-shields these protons causing them to appear downfield when compared to alkane protons. Protons on carbon adjacent to the alcohol oxygen show up in the region of 3.4-4.5 ppm.In addition, the IR spectra will show the bands typical for aromatic compounds in the region of 1500-1600 cm -1. The IR spectrum of phenols the O-H stretch appears at roughly 3500 cm -1. The broadness of the O-H peak makes it very easy to distinguish in an IR spectrum. 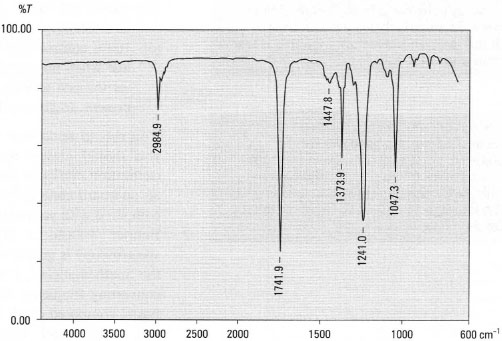
Instead of seeing one sharp peak, you see a broad set of multiple overlapping peaks. Because protons are shared to varying extent with neighboring oxygens, the covalent O-H bonds in a sample of alcohol all vibrate at slightly different frequencies and show up at slightly different positions in the IR spectrum. The rounded shape of most O-H stretching modes occurs because of hydrogen bonding between different hydroxy groups. Peak shapes are sometimes very useful in recognizing what kind of bond is present. Source: SDBSWeb : (National Institute of Advanced Industrial Science and Technology of Japan, 14 July 2008) 
In the IR spectra of 1-butanol, show below, the O-H stretch appears at 3300 cm -1 and the C-O stretch appears at 1073 cm -1.įigure IR8. In addition alcohol have a strong C-O stretch near 1000 cm -1. This exact position of the peak is dependent on the amount of hydrogen bonding in the alcohol. This peak tends to be very strong and very broad. The IR spectrum of aliphatic alcohols have a distinctive O-H stretch in the range of 3300 to 3400 cm -1. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
